
免疫性血小板减少症(Immune Thrombocytopenia, ITP)曾被称为“特发性血小板减少性紫癜”,是一种由于获得性免疫功能紊乱导致的自身免疫性疾病,临床主要表现为皮肤黏膜出血,严重者可发生颅内出血。的临床共识已将其定义为一种异质性极强的综合征,而非单一疾病。在流行病学层面,随着诊断技术的精确化,成人ITP的发病率在不同性别与年龄段中表现出明显的双峰分布,这暗示了不同年龄阶段可能存在差异化的免疫致病驱动力。
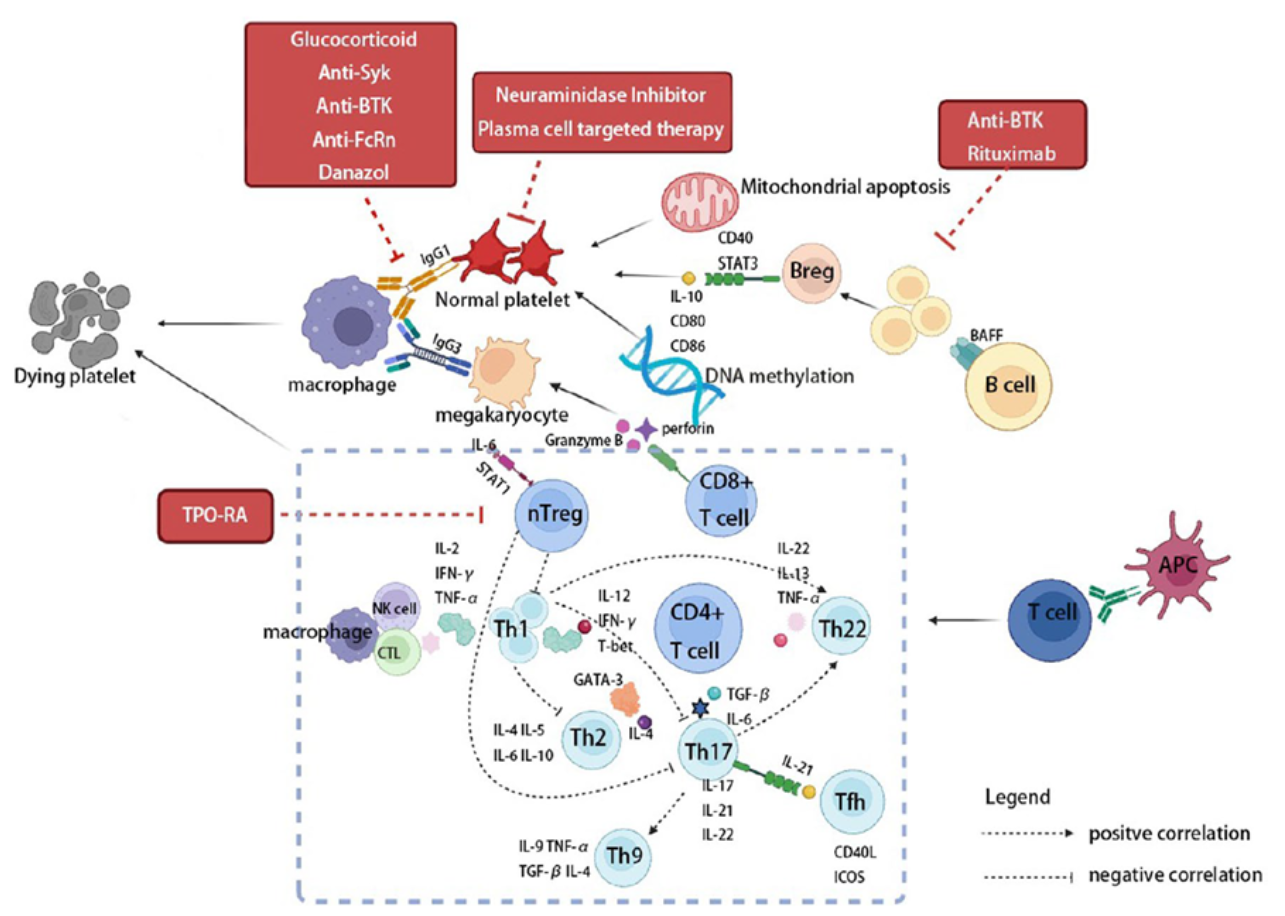
Figure 1. ITP发病机制和新疗法的简化示意图
ITP的致病机制复杂,涉及体液免疫、细胞免疫及巨核细胞功能障碍等多环节。患者体内产生针对血小板糖蛋白的IgG自身抗体,这些抗体通过Fcγ受体介导的吞噬作用促使脾脏和肝脏巨噬细胞清除血小板;同时,抗体还可诱导血小板去唾液酸化,暴露N-乙酰氨基葡萄糖残基,进一步被肝脏Ashwell-Morell受体识别清除。自身抗体亦可结合骨髓巨核细胞,抑制其成熟和血小板生成,导致血小板生成不足。细胞免疫层面,调节性T细胞(Treg)和B细胞(Breg)功能缺陷,Th1/Th17亚群优势,细胞毒性CD8?T细胞通过穿孔素/颗粒酶B途径直接诱导血小板和巨核细胞凋亡;B细胞活化因子(BAFF)过度表达进一步促进自身反应性B细胞存活和抗体产生。补体激活及表观遗传异常(如DNA甲基化改变)也参与其中,形成“破坏增加+生成减少”的双重病理。
近年来ITP治疗进入精准免疫调节新时代。除传统糖皮质激素、IVIG、利妥昔单抗(rituximab)和血小板生成素受体激动剂(TPO-RAs,如eltrombopag、romiplostim)外,多项新型药物已进入临床应用或III期试验。
BTK(Bruton酪氨酸激酶)是B细胞受体(BCR)和Fcγ受体(FcγR)信号通路下游的关键激酶。2025年,口服可逆共价BTK抑制剂rilzabrutinib(商品名Wayrilz)获FDA批准,成为针对ITP的BTK抑制剂。该药通过选择性抑制BTK激酶(C481位点可逆共价结合),多途径调控免疫反应:一方面阻断B细胞受体(BCR)下游信号,抑制自身反应性B细胞活化及自身抗体产生;另一方面阻断Fcγ受体(FcγR)介导的巨噬细胞吞噬作用,减少抗体包被血小板的清除;同时保留血小板G蛋白偶联受体(如CLEC-2)介导的功能,避免增加出血风险。LUNA 3 III期试验显示,在既往治疗失败的慢性ITP患者中,rilzabrutinib实现快速且持久的血小板升高,应答率显著优于安慰剂,且安全性良好。
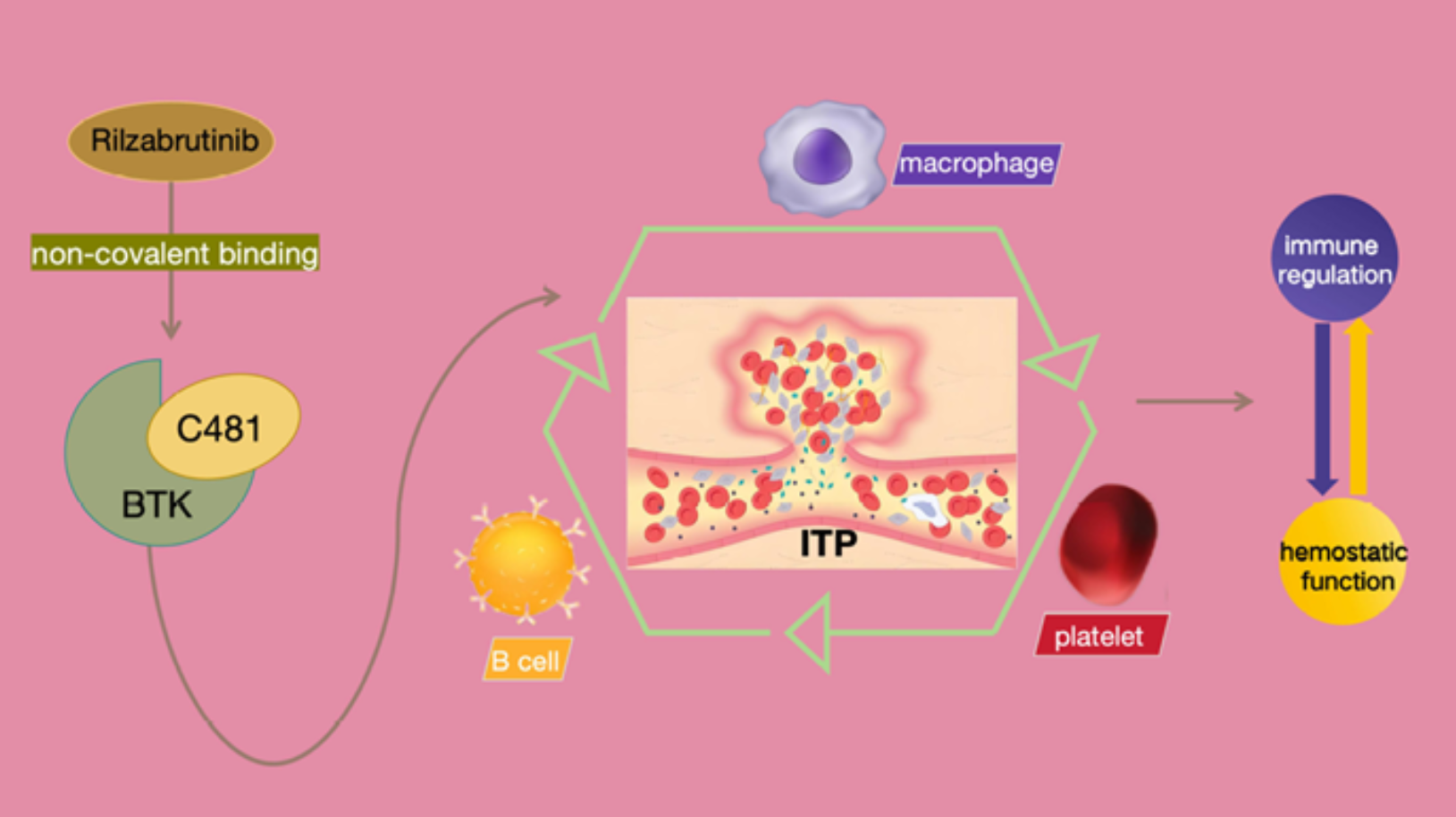
Figure 2. Rilzabrutinib在ITP中的作用机制和临床转化示意图
FcRn(新生儿Fc受体)是调控IgG稳态的关键受体,通过酸性内体环境中结合IgG防止其溶酶体降解。FcRn拮抗剂efgartigimod(工程化IgG1 Fc片段)以高亲和力竞争性结合FcRn,阻断IgG(包括致病性自身抗体)的再循环,促进其降解,从而特异性降低循环总IgG水平(通常60-80%)。ADVANCE IV III期试验显示,efgartigimod显著提高慢性ITP患者的持续血小板应答比例(≥50×10?/L在规定访视中≥4/6次),改善出血症状和生活质量,目前开放标签扩展研究正在评估长期疗效。
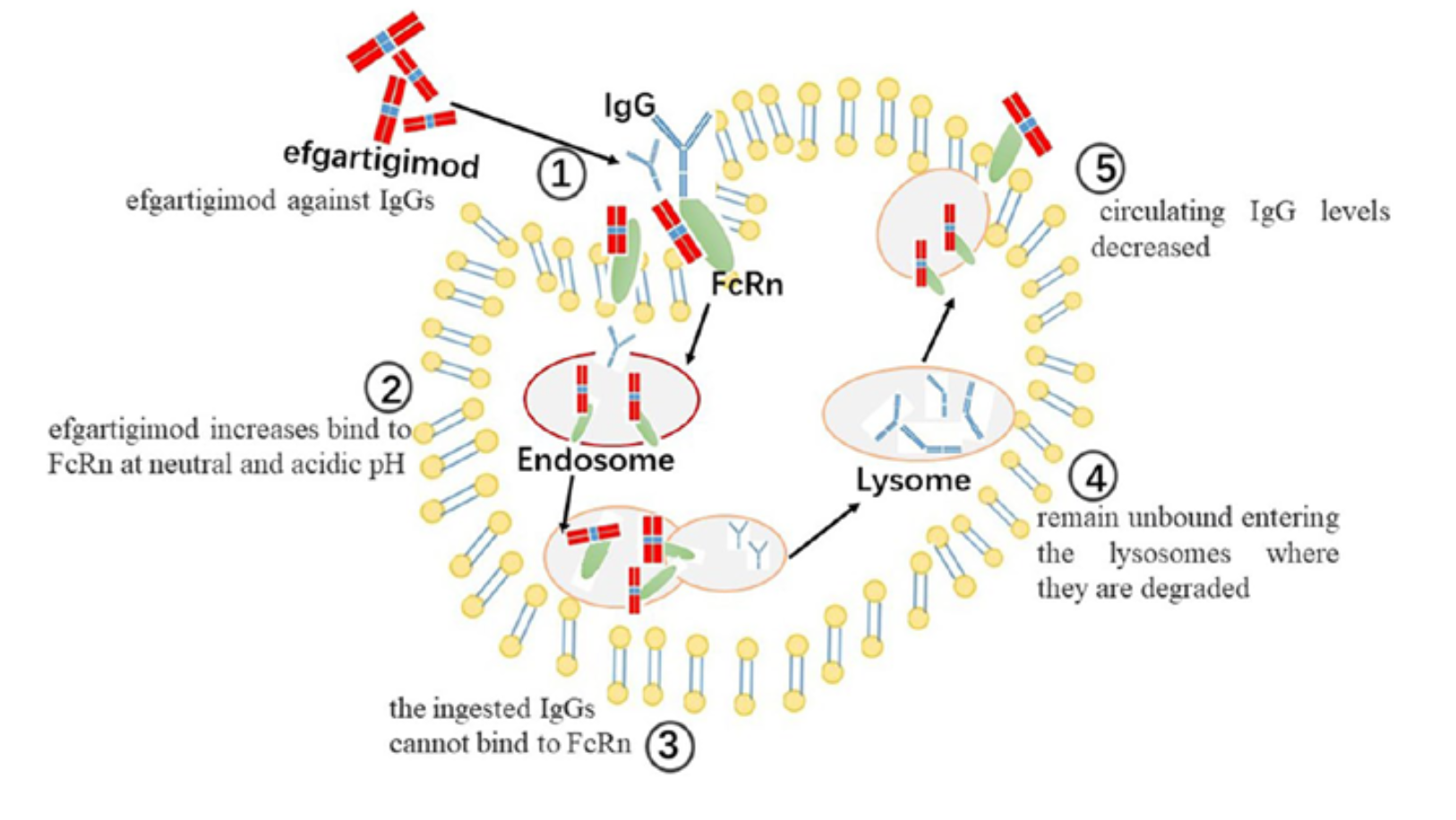
Figure 3. efgartigimod的药理作用示意图
CD38在产生自身抗体的长寿浆细胞表面高表达。抗CD38单抗(如mezagitamab、CM313、daratumumab)通过抗体依赖性细胞毒性(ADCC)、补体依赖性细胞毒性(CDC)及直接凋亡诱导等机制,特异性耗竭浆细胞,从源头减少抗体生成。CM313 II期随机对照试验及daratumumab II期研究显示,在难治性ITP患者中可实现快速血小板计数恢复并维持应答;mezagitamab II期试验也显示类似快速持久效果,目前相关III期研究正在推进。
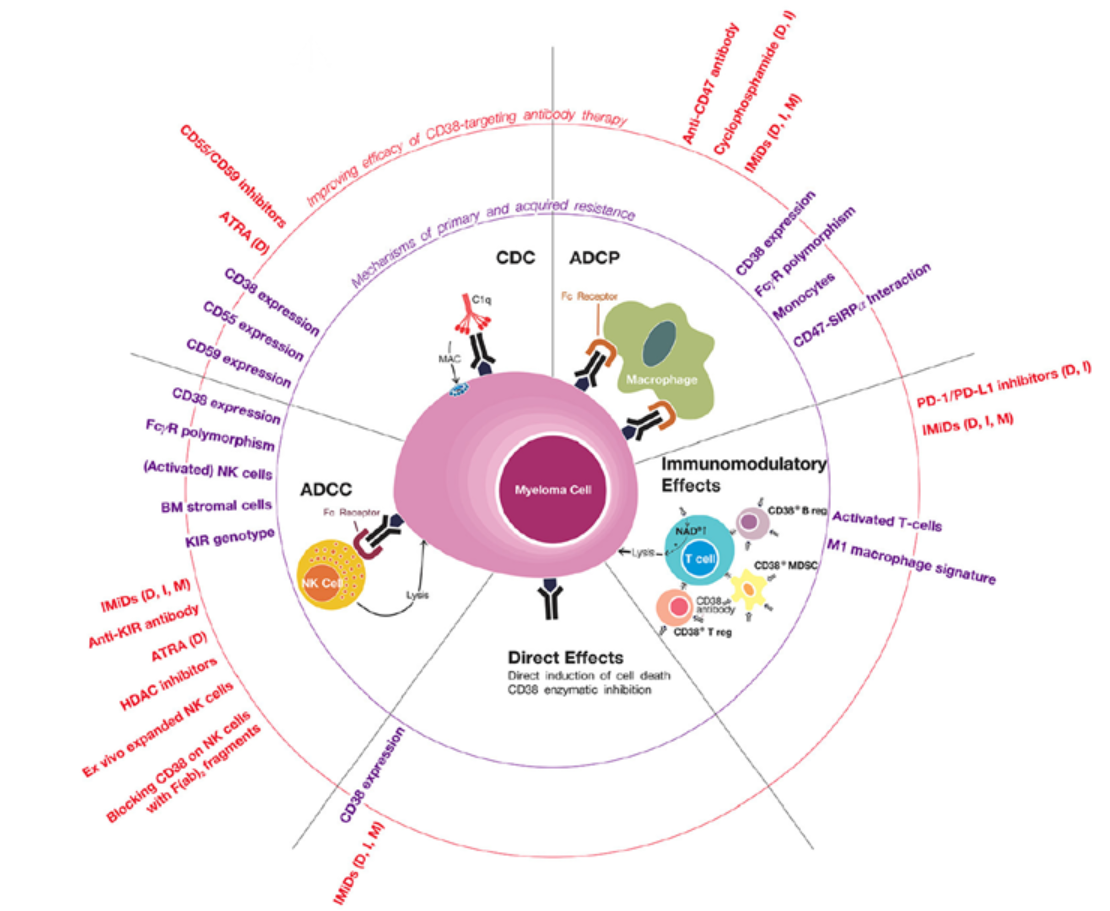
Figure 4. 对CD38抗体的原发性和获得性抗性的机制
Syk(脾酪氨酸激酶)是FcγR信号通路上游关键节点。Syk抑制剂fostamatinib(商品名Tavalisse,已于2018年获FDA批准)通过抑制Syk,阻断巨噬细胞对IgG包被血小板的吞噬作用,同时抑制B细胞活化及抗体产生。FIT临床试验程序证实其在慢性ITP中显著提高血小板应答率。
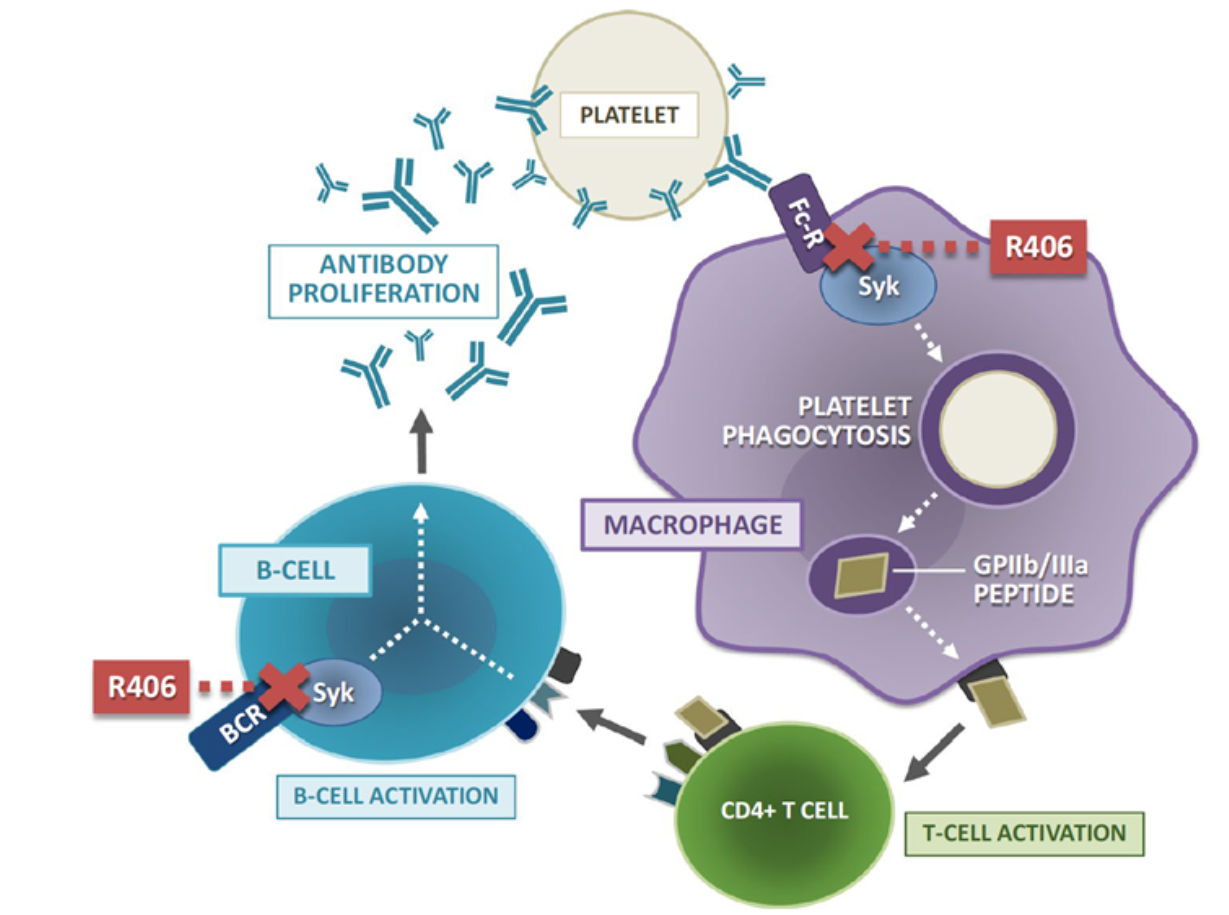
Figure 5. fostamatinib活性代谢物R406在免疫性血小板减少症作用机制示意图
BAFF-R(B细胞活化因子受体)调控B细胞存活、成熟和增殖。BAFF-R抑制剂ianalumab(VAY736,全人源IgG1单抗)通过阻断BAFF-R信号并增强ADCC介导的B细胞耗竭,抑制自身反应性B细胞功能。VAYHIT2 III期试验结果显示,在一线糖皮质激素治疗失败的原发性ITP患者中,ianalumab联合eltrombopag较安慰剂联合组显著延长治疗失败时间(TTF),提高6个月稳定应答率,并有助于减少eltrombopag用量,具有潜在疾病修饰作用。
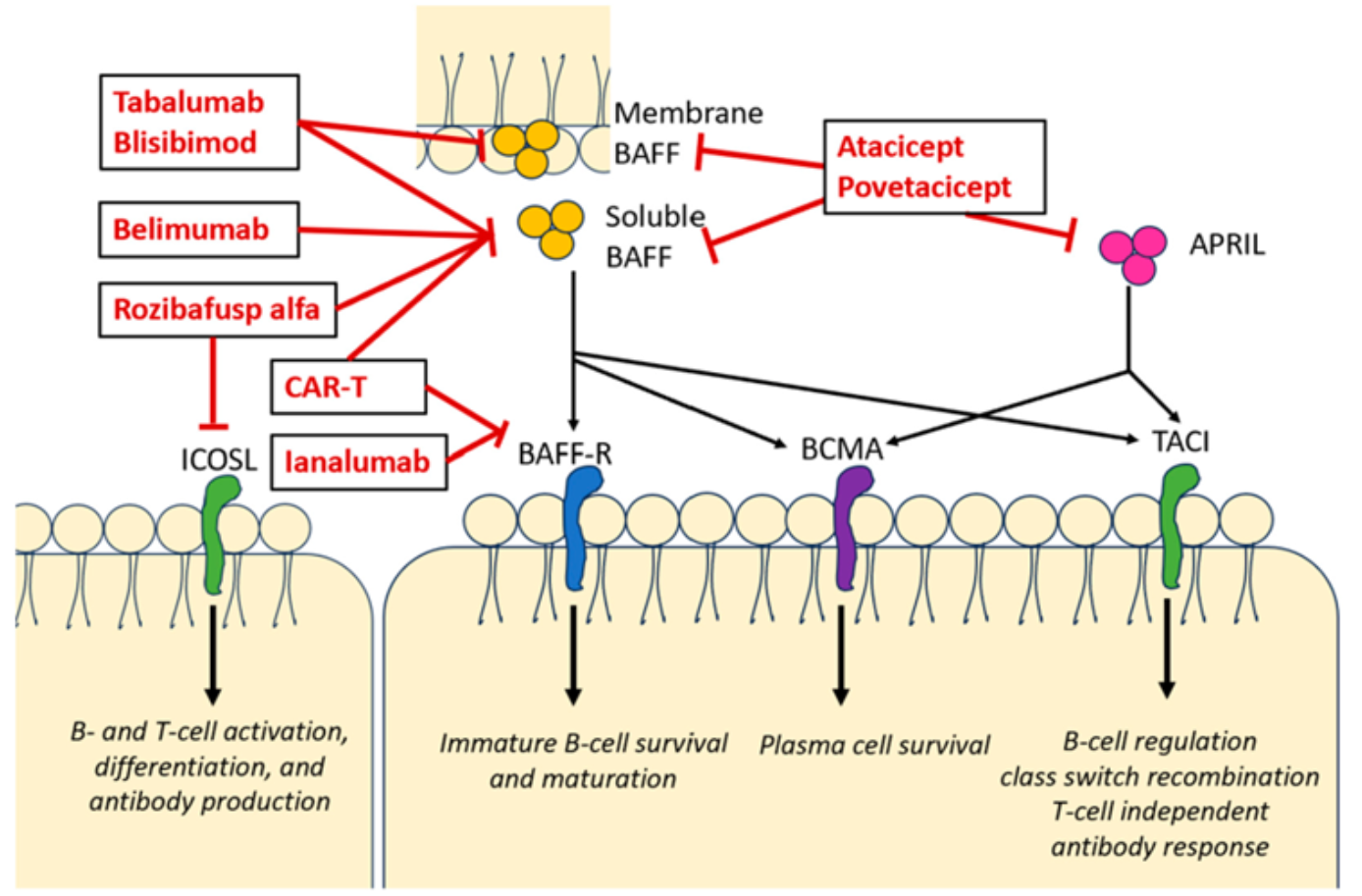
Figure 6. 抗BAFF药物的作用机制
这些精准靶向药物共同构成了ITP治疗的新时代框架,未来多靶点联合或基于个体免疫表型的方案有望进一步优化患者预后。临床应用中需关注各药物的适应证、监测指标及安全性特征。
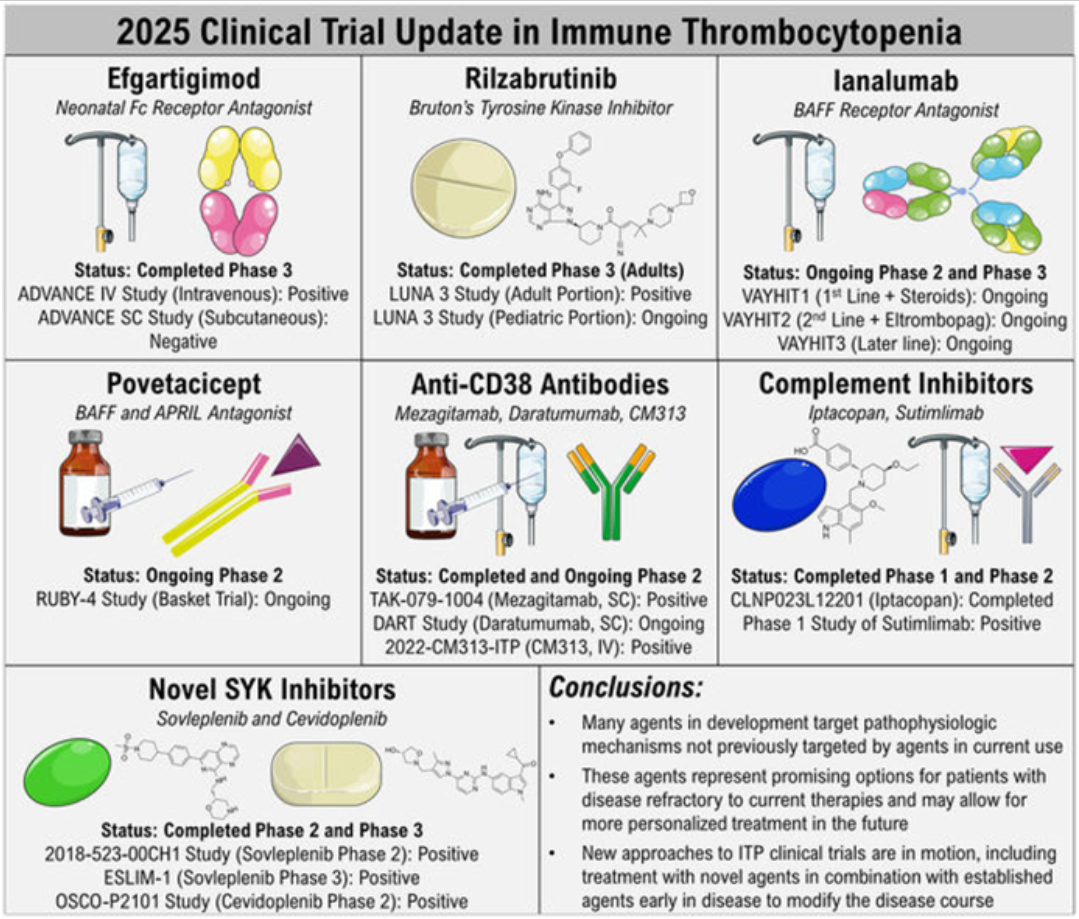
Figure 7. 2025年免疫性血小板减少症临床试验更新
以下为 abinScience 针对免疫性血小板减少症相关的重组蛋白与抗体产品,目录号可直接点击进入产品页。
| Catalog No. | Product name |
| HT068012 | Recombinant Human PIK3CD Protein, N-His |
| HF996011 | Recombinant Human CD25/IL2RA Protein, C-His |
| HY477012 | Recombinant Human CD42b/GP1BA Protein, N-His |
| HY386022 | Recombinant Human CD16a/FCGR3A Protein, N-His |
| HY598012 | Recombinant Human C1S Protein, N-His |
| HY598011 | Recombinant Human C1S Protein, C-His |
| HY257012 | Recombinant Human CD20/MS4A1 Protein, N-His |
| HY139012 | Recombinant Human CD32/FCGR2A Protein, N-His |
| HB990012 | Recombinant Human CD119/IFNGR1 Protein, N-His |
| HB011012 | Recombinant Human CD22 Protein, N-His |
| Catalog No. | Product name |
| HX061016 | Research Grade Orilanolimab |
| HX061026 | Research Grade Rozanolixizumab |
| HX061086 | Research Grade imeroprubart |
| HV212026 | Research Grade Tabalumab |
| HV212016 | Research Grade Belimumab |
| HB199016 | Research Grade Letolizumab |
| HB199026 | Research Grade Dapirolizumab |
| HB199096 | Research Grade Tegoprubart |
| HY598016 | Research Grade Sutimlimab |
| HY598026 | Research Grade Riliprubart |
| Catalog No. | Product name |
| DF996018 | Basiliximab ELISA Kit |
| DF996028 | Camidanlumab ELISA Kit |
| DF996038 | Daclizumab ELISA Kit |
| DY257018 | Obinutuzumab ELISA Kit |
| DY257028 | Afutuzumab ELISA Kit |
| DY257058 | Ibritumomab ELISA Kit |
| DY257078 | Ocaratuzumab ELISA Kit |
| AY257028 | Anti-Obinutuzumab ELISA Kit |
| AY257018 | Anti-Rituximab ELISA Kit |
| DB199018 | Letolizumab ELISA Kit |
需要ITP研究工具?
abinScience提供原厂直供的免疫性血小板减少症相关重组蛋白、抗体及ELISA试剂盒,支持批量询价、定制规格及技术咨询。
立即询价
联系邮箱:info@abinscience.com | 电话:027--87433958
Reference:
1. Cheekati M, Murakhovskaya I. Anti-B-Cell-Activating Factor (BAFF) Therapy: A Novel Addition to Autoimmune Disease Management and Potential for Immunomodulatory Therapy in Warm Autoimmune Hemolytic Anemia. Biomedicines. 2024 Jul 18;12(7):1597. doi: 10.3390/biomedicines12071597. PMID: 39062171; PMCID: PMC11275058.
2. Neunert CE, Arnold DM, Grace RF, Kuhne T, McCrae KR, Terrell DR. The 2022 review of the 2019 American Society of Hematology guidelines on immune thrombocytopenia. Blood Adv. 2024 Jul 9;8(13):3578-3582. doi: 10.1182/bloodadvances.2023012541. PMID: 38608258; PMCID: PMC11319830.
3. Liu L, Xiao Y, Jia Y, Shao Z, Shi J, Cui C. From C481 Resistance Evasion to Platelet Preservation: Rilzabrutinib Redefines ITP Targeted Therapy. Drug Des Devel Ther. 2025 Sep 11;19:8161-8180. doi: 10.2147/DDDT.S543620. PMID: 40959738; PMCID: PMC12435525.
4. Yang Y, Shen Z, Shi F, Wang F, Wen N. Efgartigimod as a novel FcRn inhibitor for autoimmune disease. Neurol Sci. 2024 Sep;45(9):4229-4241. doi: 10.1007/s10072-024-07460-5. Epub 2024 Apr 22. PMID: 38644454.
5. van de Donk NWCJ and Usmani SZ (2018) CD38 Antibodies in Multiple Myeloma: Mechanisms of Action and Modes of Resistance. Front. Immunol. 9:2134. doi: 10.3389/fimmu.2018.02134
6. Paik J. Fostamatinib: A Review in Chronic Immune Thrombocytopenia. Drugs. 2021 Jun;81(8):935-943. doi: 10.1007/s40265-021-01524-y. Epub 2021 May 10. Erratum in: Drugs. 2021 Jul;81(11):1361. doi: 10.1007/s40265-021-01540-y. PMID: 33970459.
7. Mititelu A, Onisai MC, Ro?ca A, Vl?d?reanu AM. Current Understanding of Immune Thrombocytopenia: A Review of Pathogenesis and Treatment Options. Int J Mol Sci. 2024 Feb 10;25(4):2163. doi: 10.3390/ijms25042163. PMID: 38396839; PMCID: PMC10889445.
8. Zufferey A, Kapur R, Semple JW. Pathogenesis and Therapeutic Mechanisms in Immune Thrombocytopenia (ITP). J Clin Med. 2017 Feb 9;6(2):16. doi: 10.3390/jcm6020016. PMID: 28208757; PMCID: PMC5332920.
9. Madkhali MA. Recent advances in the management of immune thrombocytopenic purpura (ITP): A comprehensive review. Medicine (Baltimore). 2024 Jan 19;103(3):e36936. doi: 10.1097/MD.0000000000036936. PMID: 38241567; PMCID: PMC10798712.
10. Al-Samkari H. 2025 update on clinical trials in immune thrombocytopenia. Am J Hematol. 2024 Nov;99(11):2178-2190. doi: 10.1002/ajh.27448. Epub 2024 Aug 6. PMID: 39105413; PMCID: PMC11469945.
11. Yan, X., Yun, Z., Tian, L. et al. Immune thrombocytopenia: a review of pathogenesis and current treatment. Discov Med 1, 57 (2024).
12. D?rner T, Posch MG, Li Y, Petricoul O, Cabanski M, Milojevic JM, Kamphausen E, Valentin MA, Simonett C, Mooney L, Hüser A, Gram H, Wagner FD, Oliver SJ. Treatment of primary Sj?gren's syndrome with ianalumab (VAY736) targeting B cells by BAFF receptor blockade coupled with enhanced, antibody-dependent cellular cytotoxicity. Ann Rheum Dis. 2019 May;78(5):641-647. doi: 10.1136/annrheumdis-2018-214720. Epub 2019 Mar 2. PMID: 30826774.
返回顶部